The H-Reflex: A Window Into the Spinal Cord Without Going Inside It
Published:
Most of what we know about the brain and spinal cord comes from indirect measurements. For decades, one of the most useful probes of spinal circuit function has been the deceptively simple H-reflex, which you can record from a muscle using just a surface electrode.
This post explains what the H-reflex is, why it reveals important information about spinal circuits, and how clinicians and researchers use it to track changes in health and disease.
What the H-reflex is, and where it came from
The H-reflex was first described by Paul Hoffmann, hence the name “H-“ reflex, in 1918 while studying the soleus muscle that controls the ankle. It is, at its core, a monosynaptic spinal reflex triggered by electrical stimulation of a nerve.
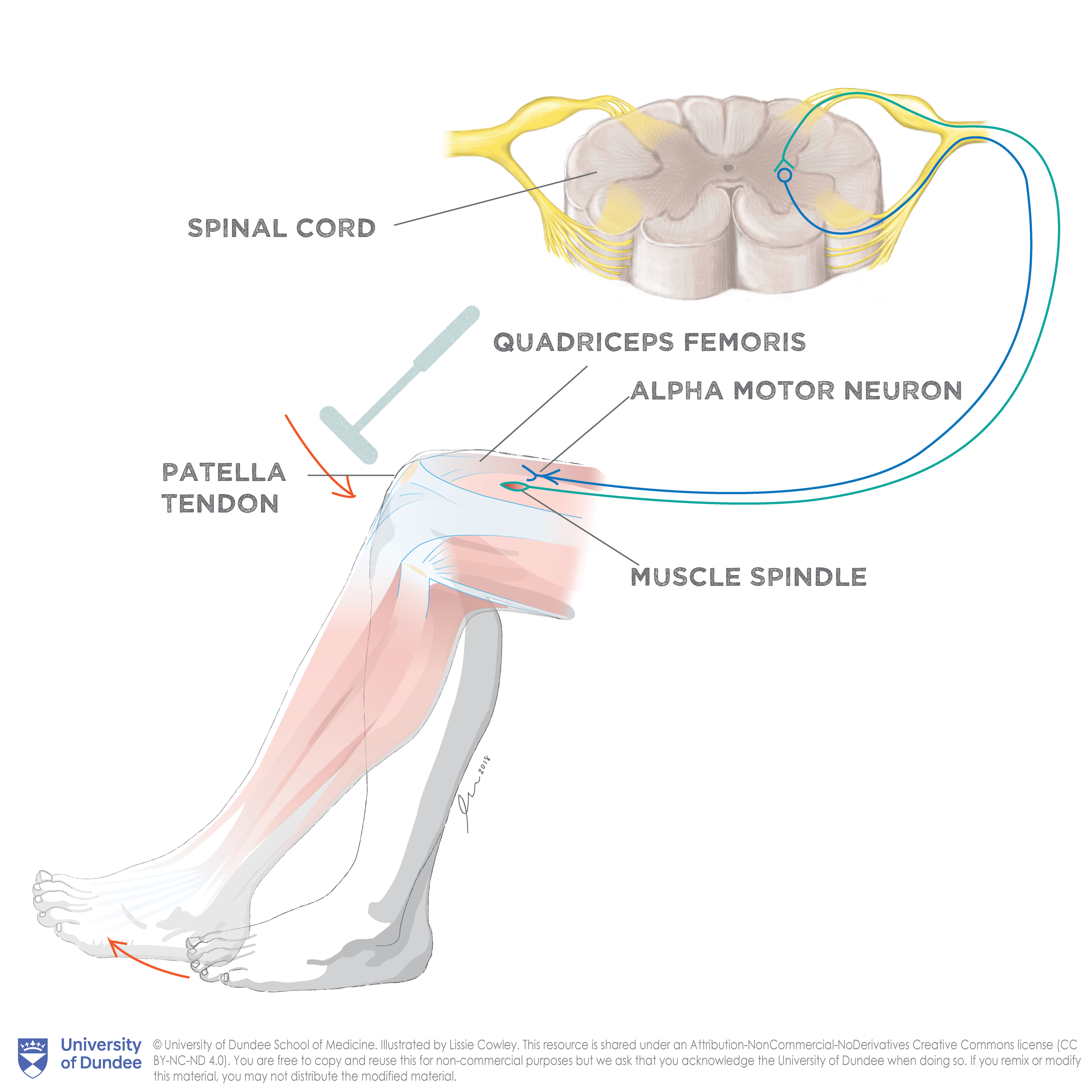
You’ve probably experienced a mechanical version of this reflex that’s commonly called the “knee-jerk” reflex. When a doctor taps your patellar tendon, the sudden stretch of your quadriceps muscle activates stretch-sensitive receptors in muscle spindles. These send electrical signals up large-diameter sensory axons (Ia afferents) to the spinal cord, where they synapse directly onto alpha motor neurons. The motor neurons then send a signal back to the muscle, producing the familiar involuntary kick.
The H-reflex uses the same neural circuit, but instead of a tendon tap, an electrical pulse is delivered to the peripheral nerve through a surface or needle electrode.
Electrical stimulation has a key advantage over the tendon tap: you can precisely control and vary the stimulus intensity. This makes the H-reflex a powerful tool for quantitative measurement, not just a yes/no diagnostic.
The recorded muscle waveform has a characteristic shape. A brief stimulus artifact marks the stimulus onset. Shortly after (about 5–11 ms, depending on limb length), the M-wave appears. This is a direct motor response from activating efferent motor axons. After a longer delay (about 25–35 ms), the H-wave arrives. This is the reflex response that traveled up the sensory pathway into the spinal cord and back down.
The recruitment curve: the shape that matters most
The most informative way to use the H-reflex is to sweep the stimulus intensity from just below threshold to the maximum response and record how both waves change. This produces a recruitment curve, whose shape encodes a lot of physiology.
At low intensities, the stimulus is too weak to recruit anything. As intensity increases:
- The H-wave appears first. Ia afferents have lower activation thresholds than motor axons, so low intensities selectively activate the sensory pathway and produce a reflex response with no direct motor activation. This is the clean window for studying the reflex arc in isolation.
- The H-wave grows, then peaks. As more afferents are recruited, more motor neurons receive synaptic input and the H-wave amplitude increases. It reaches a maximum (H-max) at moderate intensity.
- The M-wave grows. Once intensity exceeds the threshold for direct motor axon activation, the M-wave appears and grows to its own maximum (M-max).
- The H-wave shrinks and disappears. At high intensities, the direct motor response travels antidromically (backward, toward the spinal cord) along motor axons, while the H-reflex travels orthodromically (toward the muscle). The two action potentials collide and cancel, blocking the reflex. This is called antidromic collision.
The H-max/M-max ratio (H/M Ratio) is a standard metric. It normalizes the maximum reflex response to the maximum direct motor response, providing a measure of reflex excitability that is independent of electrode placement or body size. The H/M ratio is therefore widely used as a biomarker of motor neuron excitability and spinal circuit function.
Interactive demo
The synthetic dataset below mirrors human H-reflex EMG recruitment curves. Explore how the H-reflex and M-wave change with stimulus intensity. Step through the 35 sweeps with the buttons, drag the slider, or click any point on the recruitment curve to jump to that sweep. Keyboard arrow keys also work.
About this data
The H-reflex is a spinal monosynaptic reflex used to assess the excitability of the motor neuron pool. As stimulus intensity increases from threshold, two distinct responses emerge in the EMG:
- M-wave — direct motor response (~5–11 ms post-stimulus). Amplitude follows a sigmoid: sub-threshold, then rapid recruitment, then saturation (M-max). The blue shaded region marks the analysis window.
- H-wave — H-reflex response via spinal cord (~24–32 ms post-stimulus). Amplitude follows an inverted-U: peaks around 3–5 mA, then disappears as antidromic M-wave collision blocks it at high intensities. The green shaded region marks the analysis window.
- Stimulus onset — the electrical pulse delivered at 5 ms, marked by the dashed red line. A brief artifact is visible immediately after.
Synthetic data generated using physiologically calibrated parameters; filtered with a 4th-order Butterworth bandpass (100–3500 Hz). 35 sweeps, 1200 samples each at 30 kHz.
Why this is useful in human health
Spasticity and “upper motor neuron” injury
Spasticity — the muscle stiffness and hyperreflexia seen after stroke, spinal cord injury, traumatic brain injury, or multiple sclerosis — involves changes in spinal inhibitory circuits. The H/M ratio is typically elevated in spastic muscles compared to the unaffected limb or healthy controls, because spinal inhibition that normally suppresses the reflex arc is reduced or lost after damage to descending motor pathways.
Quantifying the H/M ratio gives clinicians a physiological measure of spasticity severity that is more specific than clinical scales. It is also used to evaluate treatment responses: baclofen (which enhances presynaptic inhibition of Ia afferents) and other antispasticity agents measurably reduce the H/M ratio.
Peripheral neuropathy
In diabetic peripheral neuropathy and other conditions that damage large sensory axons, the H-reflex is often one of the first electrophysiological findings to become abnormal. Ia afferents are large and myelinated, making them especially vulnerable to damage from metabolic or mechanical causes. In patients with degenerating Ia afferents, the H-reflex may show prolonged latency (slowed conduction) or be absent altogether in advanced cases. This makes it a useful complement to nerve conduction studies in early neuropathy screening.
Radiculopathy
The most common clinical use of the H-reflex is diagnosing S1 nerve root compression. The tibial nerve H-reflex predominantly tests the S1 reflex arc, so a unilateral absence or prolonged latency is a sensitive marker of S1 radiculopathy (e.g., from a herniated L5-S1 disc). It is more sensitive than the Achilles tendon reflex on physical exam, especially in older patients whose ankle jerk may be diminished at baseline.
Drug effects
Because the H-reflex loop is entirely spinal, drugs that act on spinal interneurons and Ia synapses have measurable effects on it. Alcohol and benzodiazepines suppress the H-reflex (consistent with their GABA-A receptor mechanisms), while caffeine and some noradrenergic agents increase it. The H-reflex is used in pharmacology studies to demonstrate target engagement of drugs designed to modulate spinal inhibition.
What the H-reflex reveals about spinal circuit organization
The reflex arc itself is straightforward, but what makes the H-reflex scientifically powerful is that you can condition it — apply a second stimulus or sensory input just before the test pulse and observe how the reflex is suppressed or facilitated. Each conditioning paradigm probes a different interneuronal circuit. Here are two examples from my own work:
Reciprocal inhibition
If you condition the soleus H-reflex with a stimulus to the common peroneal nerve (which activates the soleus’s antagonist, the tibialis anterior), the H-reflex is briefly suppressed about 1–3 ms later. This is called Ia reciprocal inhibition: the conditioning stimulus activates Ia inhibitory interneurons that inhibit soleus motor neurons. These are the same interneurons that mediate the spinal reciprocal inhibition underlying normal movement. The strength and timing of this suppression can be measured precisely using the H-reflex. In spasticity, reciprocal inhibition is reduced, meaning the antagonist fails to suppress the reflex as it should, thus contributing to co-contraction and stiffness.
Recurrent inhibition (Renshaw cells)
Renshaw cells receive excitatory input from motor‑axon collaterals and provide inhibitory feedback onto the same and synergist motor neurons. Anatomically, they are well characterized, but their functional role in ongoing motor control is not fully understood and is likely context dependent. They may stabilize motor‑neuron firing, shape reflex gain, and/or participate in rhythm generation, with effects that vary by species, muscle, and behavioral state. You can probe recurrent inhibition with paired H‑reflex conditioning.
A typical test gives a short conditioning pulse that creates a backward (antidromic) motor signal just before the test pulse. If the H‑wave is quickly reduced after the conditioning pulse, that points to recurrent (postsynaptic) inhibition from Renshaw cells. To check whether the change is happening at the nerve ending instead, watch the M‑wave: if the M stays the same but the H gets smaller, that suggests a central spinal effect. Antidromic conditioning can also cause peripheral collisions or temporary inability of axons to fire, so use simple controls (e.g., vibration or stimulating a different nerve), try different conditioning intervals, and average many trials to be confident where the inhibition is coming from.
In short: conditioning the H‑reflex can show Renshaw‑mediated suppression, but simple controls are needed to rule out peripheral or presynaptic effects.
The limits of the method
In humans, the H-reflex is easy to record in the soleus, flexor carpi radialis (forearm muscle), and a few other muscles. In most muscles, however, it is not recordable — the anatomy is inaccessible, or the signal is buried in noise from nearby muscles. This limits which spinal circuits you can study.
More fundamentally, the H-reflex is a population measure. Its amplitude reflects the net state of hundreds of motor neurons, dozens of interneuronal circuits, and the entire Ia afferent population all averaged together. It cannot tell you which specific interneuron population is abnormal; it only tells you the aggregate output. Conditioning paradigms help dissect contributions, but interpretation always involves assumptions about the underlying circuitry that are in some cases still debated.
Finally, it is not a static measurement. The H-reflex changes with posture, attention, arousal, recent voluntary movement, time of day, and even breath holding. Running a reliable protocol requires controlling many variables and averaging across many trials to get a stable estimate of reflex excitability.
The recruitment curve as a data structure
One underappreciated aspect of H-reflex methodology is how much information is encoded in the full recruitment curve—not just H-max/M-max. The stimulus intensity at which the H-wave first appears, the steepness of its rising limb, the intensity at which it begins to decline, and the ratio of H-max to the M-wave amplitude at H-max all reflect different aspects of motor neuron pool excitability, Ia afferent thresholds, and synaptic input balance.
In my own work, I use a custom program I made (MonStim Analyzer) to extract interesting features from the recruitment curve for each of my datasets. The interactive demo here in this post shows what the underlying data look like for a typical adult human: a stimulus intensity sweep with waveform and recruitment curve displayed together, using the same signal processing pipeline that my full application uses (Butterworth bandpass filter, RMS and peak-to-trough amplitude extraction).
Summary
The H-reflex is just an EMG signal — a few millivolts recorded from a surface electrode. But the circuit that generates it passes throughashrough the spinal cord, making it a surprisingly rich probe of spinal physiology. The reflex recruitment curve reveals motor neuron excitability and samples the integrity of the sensory-motor loop. Conditioning paradigms let you interrogate specific inhibitory and excitatory interneuronal circuits without recording from a single spinal neuron. Clinical applications span spasticity, neuropathy, radiculopathy, and pharmacology, and with the advent of surface EMG and portable stimulators, it is more accessible and less invasive than ever for both research and clinical use.
Ultimately, the H‑reflex endures because it is practical. No implants, no surgery — just a stimulator, a couple of electrodes, and an amplifier. You can set up a protocol in minutes. For investigators and clinicians alike it remains a dependable, minimally invasive assay of human spinal circuitry. Its limitations — a population‑level signal, sensitivity to posture and arousal, and restricted muscle availability — are counterbalanced by the depth of insight it can provide when experiments are rigorously designed and interpreted. As electrophysiology continues its march toward more mobile and wearable systems, the H‑reflex will likely stay in our toolbox.
